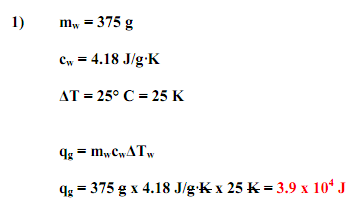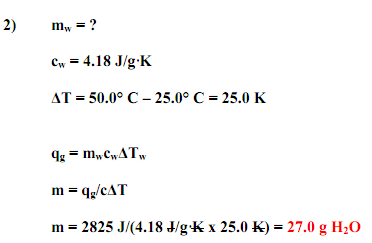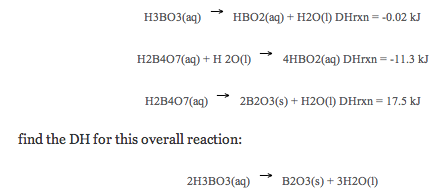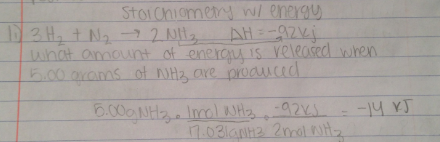Chapter 10
Understand Energy
Energy produces heat, is the ability to do work, and ability to move a substance
Types of Energy: Potential Energy and Kinetic Energy(Energy of motion)
Potential Energy Types: Elastic, Gravitational(goes against gravity), and Chemical(energy is stored in bonds-break bonds to release energy)
Understand the Law of Conservation of Energy
The Law of Conservation of Energy is that energy cannot be created not destroyed only transferred to another form
Understand Temperature and Heat
Temperature is the measurement of energy which measures random motion(kinetic energy)
Heat is the transfer of energy, flow of energy due to a temperature difference
goes from hot to cold (low to high)
Understand Exothermic and Endothermic
Exothermic- heat goes out (leaves system)
Endothermic- heat goes in system
Understand Thermodynamics
the study of energy
1st law of thermodynamics- energy of the universe is constant
Internal energy (energy of the system)
Understand and calculate specific heat Capacity and energy
Specific Heat Capacity is the amount of energy required to change the temperature of one gram of a substance by one Celsius degree
Calculation- Q= s X m X Δ T
where Q= energy(heat) required
s= specific heat capacity
m= mass of the sample in grams
Δ T= change in temperature in Celsius degrees
Calculating energy involves stoichiometry
You must find how much energy is released by doing stoichiometric conversions and converting to KJ
Understand Enthalpy
Enthalpy is the flow of heat
Understand and Calculate Hess’s Law
The principle, which is known as Hess's Law, can be illustrated by examining the oxidation of nitrogen to produce nitrogen dioxide.
Understand energy as a driving force for chemical reactions
In many years of of analyzing many processes, scientists have discovered two very important driving forces
Energy Spread and Matter Spread
Energy Spread means that in a given process, concentrated energy is dispersed widely. This distribution happens every time an exothermic process occurs.
Matter spread means exactly what it says: the molecules of a substance are spread out and occupy a larger volume.
Entropy is a function we have intended to keep track of the natural tendency for the components of the universe to become disordered-entropy is a measure of disorder of randomness
A spontaneous process is one that occurs in nature without outside intervention.
The second law of thermodynamics helps us to understand why certain processes are spontaneous and others are not.
It also helps us to understand the conditions necessary for a process to be spontaneous.
Energy produces heat, is the ability to do work, and ability to move a substance
Types of Energy: Potential Energy and Kinetic Energy(Energy of motion)
Potential Energy Types: Elastic, Gravitational(goes against gravity), and Chemical(energy is stored in bonds-break bonds to release energy)
Understand the Law of Conservation of Energy
The Law of Conservation of Energy is that energy cannot be created not destroyed only transferred to another form
Understand Temperature and Heat
Temperature is the measurement of energy which measures random motion(kinetic energy)
Heat is the transfer of energy, flow of energy due to a temperature difference
goes from hot to cold (low to high)
Understand Exothermic and Endothermic
Exothermic- heat goes out (leaves system)
Endothermic- heat goes in system
Understand Thermodynamics
the study of energy
1st law of thermodynamics- energy of the universe is constant
Internal energy (energy of the system)
Understand and calculate specific heat Capacity and energy
Specific Heat Capacity is the amount of energy required to change the temperature of one gram of a substance by one Celsius degree
Calculation- Q= s X m X Δ T
where Q= energy(heat) required
s= specific heat capacity
m= mass of the sample in grams
Δ T= change in temperature in Celsius degrees
Calculating energy involves stoichiometry
You must find how much energy is released by doing stoichiometric conversions and converting to KJ
Understand Enthalpy
Enthalpy is the flow of heat
Understand and Calculate Hess’s Law
The principle, which is known as Hess's Law, can be illustrated by examining the oxidation of nitrogen to produce nitrogen dioxide.
Understand energy as a driving force for chemical reactions
In many years of of analyzing many processes, scientists have discovered two very important driving forces
Energy Spread and Matter Spread
Energy Spread means that in a given process, concentrated energy is dispersed widely. This distribution happens every time an exothermic process occurs.
Matter spread means exactly what it says: the molecules of a substance are spread out and occupy a larger volume.
Entropy is a function we have intended to keep track of the natural tendency for the components of the universe to become disordered-entropy is a measure of disorder of randomness
A spontaneous process is one that occurs in nature without outside intervention.
The second law of thermodynamics helps us to understand why certain processes are spontaneous and others are not.
It also helps us to understand the conditions necessary for a process to be spontaneous.
This website explains thermodynamics, Energy, Heat, and Enthalpy, Specific Heat Capacity, and Hess’s Law.
This website explains the Law of Conservation of Energy.
This website explains endothermic and exothermic.
This video shows how to calculate specific heat capacity.
This video explains endothermic and exothermic.
This video helps explain Hess's Law.
This video helps to explain calculating energy.
This is a Hess's Law worksheet with practice problems.
This is a website with specific heat capacity and calculating energy practice problems.