Chapter 14
3 States of Matter:
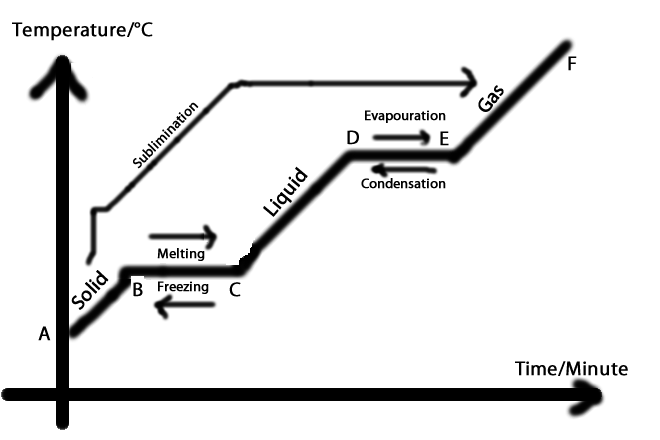
1. Solid: rigid; has a fixed shape and volume
ex. Ice-water molecules are locked into rigid positions and are close together
2. Liquid: has a definite volume but takes the shape of its container
ex. Water- water molecules are still close together but can move around to some extent
3. Gas: has no fixed volume or shape; it takes the shape and volume of its container
ex. Steam- the water molecules are far apart and move randomly
Phase Changes
1. Evaporation:
Change of phase from liquid to gas that occurs at the surface of a liquid
The temperature of any substance is related to the average kinetic energy of its particles.
Some particles have low kinetic energy and some have high kinetic energy.
When there is enough kinetic energy in a particles the particle can escape in to the atmosphere
When the molecule leaves, there is less energy in the system.
The lower kinetic energy= lower temperature
We have sweat gland in order to reduce our temperature.
Some animal like pigs and dogs do not have sweat gland.
They must find other method to cool down.
What would happen when there is equal amount of evaporation and condensation?
no change
2. Boiling
Boiling point
A temperature at which a liquid boils.
Pressure affect the boiling point
increase pressure boiling points increase
Decrease pressure boiling point decreases.
In science official boiling points is at standard pressure.
Bubbles: in liquid can form only when the pressure of the vapor with in the bubbles is great enough to resist the pressure of the surrounding liquid.
Changes liquid to gas.
Like evaporation
Major difference
Evaporation only occurs at the surface
Boiling can occur any in the liquid.
3. Condensation:
The changing of a gas to a liquid
Kinetic energy~ energy of attraction
When gas molecules near the surface of a liquid are attracted to the liquid, they strike the surface with increase kinetic energy and become part of the liquid
Condensation is a warming process
Steam burns are more painful than a burn from boiling water at the same temperature.
The steam releases considerable energy when it condenses to a liquid and wets the skin.
This release of energy is used by steam heating systems
What would happen when there is more evaporation than condensation ?
Cooling
What would happen when there is more condensation than evaporation?
increase in temperature
4. Freezing
When kinetic energy is low and the energy of attraction is greater, a liquid will freeze
Different liquids have different freezing points
5. Melting
If the kinetic energy is high enough to equal to energy of attraction then the solid becomes a liquid
Fog and Clouds
Warm air rises and expand.
When the air expand it is chills and the water molecules will slow down and stick together.
Clouds and fogs are form when lots of molecules stick together.
Forces
Intramolecular Forces: forces within in molecule
-Covalent
-Ionic
Intermolecular Forces: forces between molecules
Types of intermolecular forces:
Dipole-dipole: forces between dipoles (a molecule in which a concentration of positive electric charge is separated from a concentration of negative charge)
hydrogen bonding: the electromagnetic attractive interaction between polar molecules in which hydrogen (H) is bound to a highly electronegative atom
London dispersion forces: a temporary attractive force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles
molar heat of fusion
H20=6.02 kJ/mol
molar heat of vaporization
H20=40.6kJ/mol
calculate energy needed to melt 8.5g of ice at 0 degrees C
-8.5g H20 * (1 mol H20)/(8.0148g H20) * (6.02 kJ)/(1 mol H20) = 2.8 kJ
Vapor Pressure
In a sealed container, a given amount of liquid in a container will decrease slightly.
Eventually, the volume will be come constant.
the rate of evaporation and condensation will equal.
Sublimation: The process of changing from a solid to a gas without passing through an intermediate liquid phase.
Deposition: The process of changing from a gas to a solid without passing through an intermediate liquid phase.
Crystalline Solids
1. Ionic Solids: are stables substances with high melting points that are held together by strong forces existing between oppositely charge ions.
2. Molecular Solids: tends to melt at relatively low temperature because the intermolecular forces that exist among the molecules are relatively weak. If the molecules has a dipole moment, then the dipole-dipole forces hold the solids together. If the molecules is non-polar, then london dispersion forces holds the solids together.
3. Atomic Solids
components are atoms
Alloys
A substance that contains a mixture of elements and has metallic properties
Substitutional Alloy: consist of a solid solution of metals
Interstitial Alloy: a compound that is formed when an atom of sufficiently small radius sits in an interstitial “hole” in a metal lattice
1. Solid: rigid; has a fixed shape and volume
ex. Ice-water molecules are locked into rigid positions and are close together
2. Liquid: has a definite volume but takes the shape of its container
ex. Water- water molecules are still close together but can move around to some extent
3. Gas: has no fixed volume or shape; it takes the shape and volume of its container
ex. Steam- the water molecules are far apart and move randomly
Phase Changes
1. Evaporation:
Change of phase from liquid to gas that occurs at the surface of a liquid
The temperature of any substance is related to the average kinetic energy of its particles.
Some particles have low kinetic energy and some have high kinetic energy.
When there is enough kinetic energy in a particles the particle can escape in to the atmosphere
When the molecule leaves, there is less energy in the system.
The lower kinetic energy= lower temperature
We have sweat gland in order to reduce our temperature.
Some animal like pigs and dogs do not have sweat gland.
They must find other method to cool down.
What would happen when there is equal amount of evaporation and condensation?
no change
2. Boiling
Boiling point
A temperature at which a liquid boils.
Pressure affect the boiling point
increase pressure boiling points increase
Decrease pressure boiling point decreases.
In science official boiling points is at standard pressure.
Bubbles: in liquid can form only when the pressure of the vapor with in the bubbles is great enough to resist the pressure of the surrounding liquid.
Changes liquid to gas.
Like evaporation
Major difference
Evaporation only occurs at the surface
Boiling can occur any in the liquid.
3. Condensation:
The changing of a gas to a liquid
Kinetic energy~ energy of attraction
When gas molecules near the surface of a liquid are attracted to the liquid, they strike the surface with increase kinetic energy and become part of the liquid
Condensation is a warming process
Steam burns are more painful than a burn from boiling water at the same temperature.
The steam releases considerable energy when it condenses to a liquid and wets the skin.
This release of energy is used by steam heating systems
What would happen when there is more evaporation than condensation ?
Cooling
What would happen when there is more condensation than evaporation?
increase in temperature
4. Freezing
When kinetic energy is low and the energy of attraction is greater, a liquid will freeze
Different liquids have different freezing points
5. Melting
If the kinetic energy is high enough to equal to energy of attraction then the solid becomes a liquid
Fog and Clouds
Warm air rises and expand.
When the air expand it is chills and the water molecules will slow down and stick together.
Clouds and fogs are form when lots of molecules stick together.
Forces
Intramolecular Forces: forces within in molecule
-Covalent
-Ionic
Intermolecular Forces: forces between molecules
Types of intermolecular forces:
Dipole-dipole: forces between dipoles (a molecule in which a concentration of positive electric charge is separated from a concentration of negative charge)
hydrogen bonding: the electromagnetic attractive interaction between polar molecules in which hydrogen (H) is bound to a highly electronegative atom
London dispersion forces: a temporary attractive force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles
molar heat of fusion
H20=6.02 kJ/mol
molar heat of vaporization
H20=40.6kJ/mol
calculate energy needed to melt 8.5g of ice at 0 degrees C
-8.5g H20 * (1 mol H20)/(8.0148g H20) * (6.02 kJ)/(1 mol H20) = 2.8 kJ
Vapor Pressure
In a sealed container, a given amount of liquid in a container will decrease slightly.
Eventually, the volume will be come constant.
the rate of evaporation and condensation will equal.
Sublimation: The process of changing from a solid to a gas without passing through an intermediate liquid phase.
Deposition: The process of changing from a gas to a solid without passing through an intermediate liquid phase.
Crystalline Solids
1. Ionic Solids: are stables substances with high melting points that are held together by strong forces existing between oppositely charge ions.
2. Molecular Solids: tends to melt at relatively low temperature because the intermolecular forces that exist among the molecules are relatively weak. If the molecules has a dipole moment, then the dipole-dipole forces hold the solids together. If the molecules is non-polar, then london dispersion forces holds the solids together.
3. Atomic Solids
components are atoms
Alloys
A substance that contains a mixture of elements and has metallic properties
Substitutional Alloy: consist of a solid solution of metals
Interstitial Alloy: a compound that is formed when an atom of sufficiently small radius sits in an interstitial “hole” in a metal lattice
heating and cooling curve
websites
heating and cooling curve
intramolecular and intermolecular forces
energy required to change states of matter
different phase changes for different states of matter
vapor pressure
types of solids
types of bonding in solids
activities
changing states of matter
videos
intramolecular and intermolecular forces
different states of matter
vapor pressure