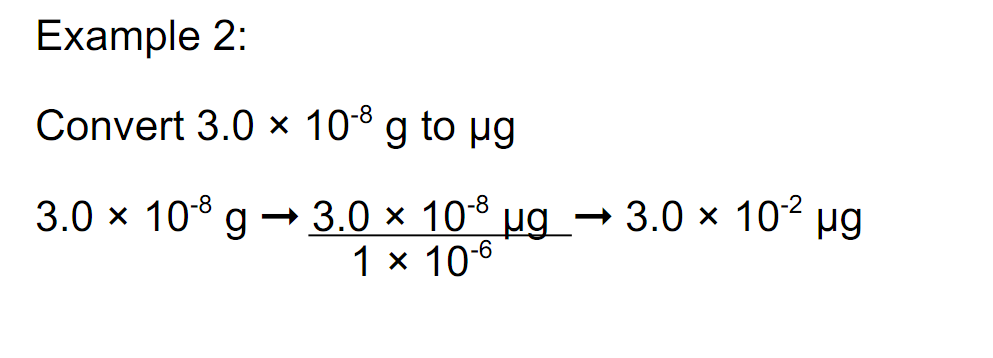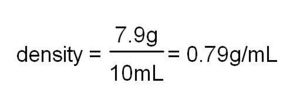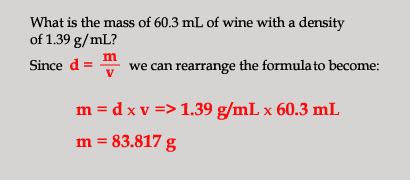Chapter 2
Scientific Notation- is a method for making very large or very small numbers more compact and easier to write.
Example: 125=1.25x100 becomes 1.25x10^2
Example: 125=1.25x100 becomes 1.25x10^2
This video is about the different units of measurement.
SI units:
http://go.hrw.com/resources/go_sc/ssp/HK1BSW25.PDF
http://www.purplemath.com/modules/metric.htm
http://www.si-units-explained.info/amount/
http://go.hrw.com/resources/go_sc/ssp/HK1BSW25.PDF
http://www.purplemath.com/modules/metric.htm
http://www.si-units-explained.info/amount/
Examples:
Uncertainty in measurement: every measurement has some degree of uncertainty
The third digit estimated and can vary
The uncertainty of a measurement depends on measuring device
http://www2.southeastern.edu/Academics/Faculty/rallain/plab194/error.html
The third digit estimated and can vary
The uncertainty of a measurement depends on measuring device
http://www2.southeastern.edu/Academics/Faculty/rallain/plab194/error.html
This video is about uncertainty of measurement.
Significant Figures: Numbers recorded in a measurement; the number of significant figures for a given measurement is determined by the inherent uncertainty of the measuring device
Examples:
0.108g=3 sf
0.0050060=5sf
Examples:
0.108g=3 sf
0.0050060=5sf
This video is about unit conversions and significant figures
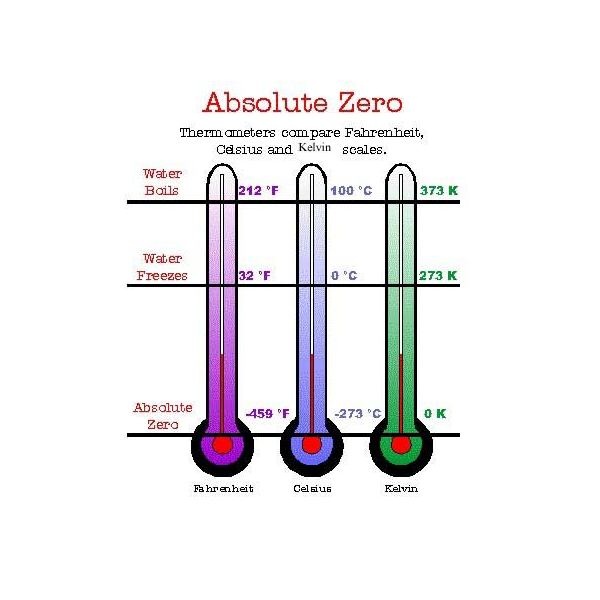
Difference between temperature scales:
Fahrenheit scale- water boils at 212 degrees and freezes at 32 degrees
Celsius scale- 0 degrees is freezing and 100 degrees is boiling
Absolute scale- water freezes at 273 K and boils at 373 K
Fahrenheit scale- water boils at 212 degrees and freezes at 32 degrees
Celsius scale- 0 degrees is freezing and 100 degrees is boiling
Absolute scale- water freezes at 273 K and boils at 373 K
Formulas:
C=(5/9) (F-32)
F=(9/5)C+32
K=273.15+C
Examples:
C= (239-32)/1.80=(207/1.80)=115
C=115
K=70.+273
K=343
F=(9/5)(100)+32
F=(900/5)+32
F=(180/1)+32
Add 180+32
F=212
C=(5/9) (F-32)
F=(9/5)C+32
K=273.15+C
Examples:
C= (239-32)/1.80=(207/1.80)=115
C=115
K=70.+273
K=343
F=(9/5)(100)+32
F=(900/5)+32
F=(180/1)+32
Add 180+32
F=212
This video is example of converting temperature scales.
Density
Density can be defined as the amount of matter present in a given volume of a substance
Formula:
Density=mass/volume
Examples:
D=(32.062g/23.50mL)=1.492g/mL
A brick of salt measuring 10.0 cm x 10.0 cm x 2.0 cm weighs 433 grams. What is its density?
Solution:
Density is the amount of mass per unit volume, or:
Density = mass/Volume
Step 1: Calculate Volume
Volume = length x width x thickness
Volume = 10.0 cm x 10.0 cm x 2.0 cm
Volume = 200.0 cm3
Step 2: Determine density
Density = mass/Volume
Density = 433 g/200.0 cm3
Density = 2.165 g/cm3
Answer:
The density of the salt brick is 2.165 g/cm3.
Density can be defined as the amount of matter present in a given volume of a substance
Formula:
Density=mass/volume
Examples:
D=(32.062g/23.50mL)=1.492g/mL
A brick of salt measuring 10.0 cm x 10.0 cm x 2.0 cm weighs 433 grams. What is its density?
Solution:
Density is the amount of mass per unit volume, or:
Density = mass/Volume
Step 1: Calculate Volume
Volume = length x width x thickness
Volume = 10.0 cm x 10.0 cm x 2.0 cm
Volume = 200.0 cm3
Step 2: Determine density
Density = mass/Volume
Density = 433 g/200.0 cm3
Density = 2.165 g/cm3
Answer:
The density of the salt brick is 2.165 g/cm3.
This video is about density.